Pauli Exclusion Principle
No two electrons in an atom can have identical quantum numbers. This is an example of a general principle which applies not only to electrons but also to other particles of half-integer spin (fermions). It does not apply to particles of integer spin (bosons). | The nature of the Pauli exclusion principle can be illustrated by
supposing that electrons 1 and 2 are in states a and b respectively. The
wavefunction for the two electron system would be but this wavefunction is unacceptable because the electrons are identical and indistinguishable. To account for this we must use a linear combination of the two possibilities since the determination of which electron is in which state is not possible to determine. |

Pauli Exclusion Principle Applications
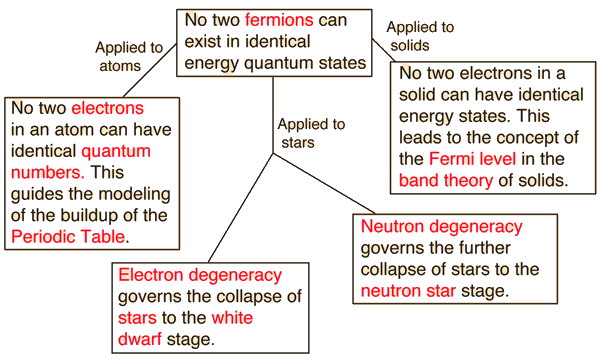
Read more...
"The Pauli Exclusion Principle states
that no two electrons can have the same four quantum numbers. We are
aware that in one orbital a maximum of two electrons can be found and
the two electrons must have opposing spins. That means one would spin up
( +1/2) and the other would spin down (-1/2)
We have the first three quantum numbers n=1, l=0, ml=0. Only two electrons can correspond to these, which would be either ms = -1/2 or ms
= +1/2. As we already know from our studies of quantum numbers and
electron orbitals, we can conclude that these four quantum numbers refer
to 1s subshell. If only one of the ms values are given then we would have 1s1 (denoting Hydrogen) if both are given we would have 1s2 (denoting Helium). Visually this would be represented as:

As you can see, the 1s subshell can hold only two electrons and when filled the electrons have opposite spins."
About (dot) com